DieselNet Technology Guide » Alternative Fuels
DieselNet | Copyright © ECOpoint Inc. | Revision 2017.09
This is a preview of the paper, limited to some initial content. Full access requires DieselNet subscription.
Please log in to view the complete version of this paper.
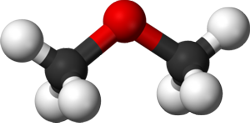
Dimethyl ether (DME) is the simplest ether, consisting of two methyl groups bonded to a central oxygen atom, as expressed by its chemical formula CH3-O-CH3, Figure 1. DME can be produced from natural gas—providing an alternative way of its utilization, in competition to such technologies as Fischer-Tropsch synthetic fuels—as well as from other carbon-containing feedstocks, including coal and various types of biomass. DME has replaced CFC gases (freons) as an environmentally friendly and safe aerosol propellant, which is one of its major current applications. Potential future uses of DME include an alternative automotive fuel, a substitute for other fuels in power generation and in the household and a source of hydrogen for fuel cells [777][3644]. Worldwide DME production grew from 100,000-150,000 tons per annum in the 1990s [153] to some 200,000 tons in the mid-2000s [1653]. China has developed a large DME production base, which reportedly amounts to almost 9 million tons per annum in installed DME capacity [1655].

With the chemical structure somewhat similar to methanol, DME contains oxygen and no carbon-carbon bonds, thus seriously limiting the possibility of forming carbonaceous particulate emissions during combustion. However, unlike methanol, DME has a high enough cetane number to perform well as a compression-ignition fuel. Also unlike methanol, DME is a gas at ambient temperature and pressure, so it must be stored under pressure as a liquid similar to LPG (liquefied petroleum gas). When used as a diesel fuel, DME provides reduced PM and NOx emissions, but increased CO and HC. Preliminary studies in the 1990s concluded that it should be possible to achieve California ULEV emissions in a passenger car using a properly designed DME fuel injection system with an HSDI engine and an oxidation catalyst [773].
The physical properties of DME (density, viscosity, lubricity, etc.) are so different from the diesel fuel that the entire fuel system must be redesigned [774]. While DME could reduce emissions compared to diesel fuel, the emission benefit comes at a price of a specific level of complexity of the fuel storage and injection system, including the need to carry a pressurized fuel tank onboard the vehicle. Furthermore, DME fuel lacks an established supply and fueling infrastructure. Therefore, the DME fuel is more likely to be used in certain niche applications, rather than provide a wide-scale alternative to liquid diesel fuels.
In an alternative utilization method, DME can be blended with propane for use in spark-ignited LPG engines. DME-LPG blends have been used in China—a country with a large DME production base—where DME has also been blended into city gas supplied for residential use.
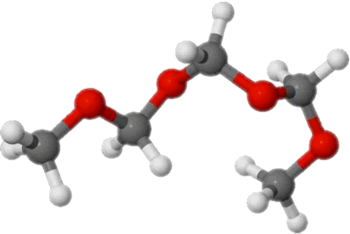
OME Fuels. Polyoxymethylene dimethyl ethers—abbreviated OME, PODE or POMDME—are a class of DME derivatives that include several oxymethylene (-O-CH2-) groups in the molecule, with a chemical formula CH3(-O-CH2)n-O-CH3 (Figure 2). OME compunds with n of 3-4 were tested as diesel engine fuels [3646]. Like DME, OME fuels are characterized by soot-free combustion.
Due to their higher molecular mass and boiling point, OME compounds may be also blended into diesel fuel. Blends of 5-10% OME in diesel were investigated in engine studies [3645]. Since OME compounds are excellent solvents, they are not necessarily compatible with the elastomers and other materials used in diesel engines.
###